Abstract
Background: In the AML1310 trial, we applied a comprehensive AML risk assessment, based on the integration of cytogenetic/genetic data and measurable residual disease (MRD) status, to optimize patients' (pts) therapeutic post-remission allocation. By doing so and using the NCCN2009 risk-stratification, favorable-risk (FR) pts (NPM1 mut/FLT3-ITD wt or CBF positive without c-Kit mutations) were to receive an autologous stem cell transplant (AuSCT); poor-risk (PR) pts (adverse karyotype or FLT3-ITD mut) were to receive an allogeneic stem cell transplant (ASCT); intermediate-risk (IR) pts (intermediate karyotype or FLT3-TKD mut or CBF positive with c-Kit mut) were to receive AuSCT or ASCT depending on the levels of MRD, measured by flow cytometry after consolidation therapy. Allocation to ASCT required the procedure to be performed whatever the source of stem cells (identical sibling, unrelated, cord blood, haploidentical). At that stage of analysis, 2-year overall (OS) and disease-free survival (DFS) of the whole series was 56% and 54%, respectively. Two-year OS and DFS were 74% and 61% in the FR category, 42% and 45% in the PR category, 79% and 61% in the IR MRD-negative category, 70% and 67% in the IR MRD-positive category (Venditti, Blood 2019:134(12);935-945) .
Aims: With an extended median follow-up of 6 years, we wanted to evaluate the long-term impact on outcome of the strategy explored in the AML1310 trial. Specifically, we meant to analyze long-term duration of OS and DFS according to the category of risk, and the corresponding post-remission therapy delivered.
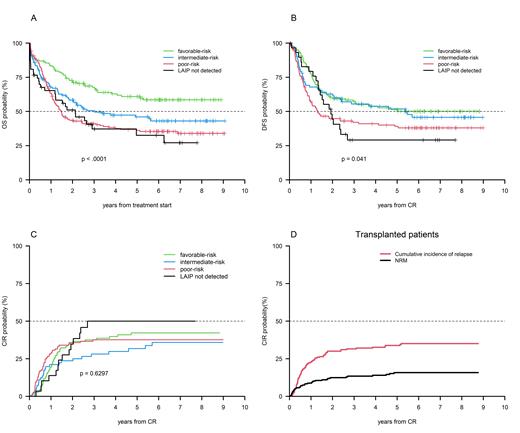
Results: Three hundred-61/500 patients (72%) achieved a CR, 342/361 completed the consolidation phase and were treatment allocated: 165 (48%) to ASCT (122 PR, 43 IR MRD-positive) plus 23 rescued after salvage therapy, for a total of 188 candidates; 150 (44%) to AuSCT (115 FR, 35 IR MRD-negative) plus 27 IR patients (8%) with no leukemia-associated phenotype, for a total of 177 candidates. Overall, 110/177 (62%) and 130/188 (71%) AuSCT or ASCT candidates received it, respectively. Median OS and DFS was 3.1 and 2.9 years, respectively. Probability of 6-years OS and DFS of the whole series was 43.6% and 43.1%, respectively; cumulative incidence of relapse (CIR) was 39.7%. Probability of 6-years OS and DFS were 58.5% and 50.1% in the FR category; 35.4% and 38.0% in the PR category; 43.1% and 45.7% in the IR category. Probability of 6-year OS and DFS of patients with no LAIP detected was 32.5% and 29.1% (Fig.1 A-B). We did not observe difference in OS and DFS between the IR-MRD negative and IR-MRD positive category: 56.6% vs 62.3% and 51.6% vs 48.6%, respectively. Six-year CIR was 42.2%, 37.6% and 35.8% in the FR, PR and IR category, respectively (Fig. 1 C). Six-year CIR of patients with no LAIP detected was 50% (Fig.1 C). Six-year CIR and non relapse mortality (NRM) of transplanted patients was 35% and 15.8%, respectively (Fig.1 D).
Conclusions: With an extended follow-up of 6 years, the present analysis confirms the long-term advantage of adopting a risk-adapted, MRD-directed strategy to implement the post-remission therapeutic decision. For patients belonging to the FR or IR-MRD negative category, an excess of toxicity was prevented by delivering an AuSCT. This is also in keeping with the ELN2017 recommendations, suggesting that AuSCT still has a role in selected situations. On the other hand, the vast majority of PR and IR-MRD positive patients received an ASCT, with a remarkable 6-year OS and DFS benefit. The effectiveness of such an approach translated in a low NRM in transplanted patients. Six-year OS, DFS and CIR of no LAIP patients also confirm that the choice of AuSCT was suboptimal and that in this situation ASCT should have been preferred. Based on the present knowledge, an MRD-directed approach is being explored also in FR patients, in the GIMEMA AML1819 trial.
Venditti: Novartis: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria, Speakers Bureau; Gilead: Consultancy, Honoraria; Astellas: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria; Jazz Pharmaceuticals: Consultancy, Honoraria, Research Funding. Luppi: Abbvie: Honoraria; Novartis: Honoraria; Sanofi: Honoraria; MSD: Honoraria; Gilead Science: Honoraria, Other: Travel grant; Daiichi-Sankyo: Honoraria; Jazz Pharma: Honoraria. Voso: Celgene/BMS: Consultancy, Honoraria, Research Funding, Speakers Bureau; Jazz: Consultancy, Honoraria, Speakers Bureau; Abbvie: Speakers Bureau; Novartis: Speakers Bureau. Vignetti: Novartis: Honoraria; Incyte: Honoraria; Amgen: Consultancy, Honoraria. Buccisano: Novartis: Honoraria, Speakers Bureau.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal